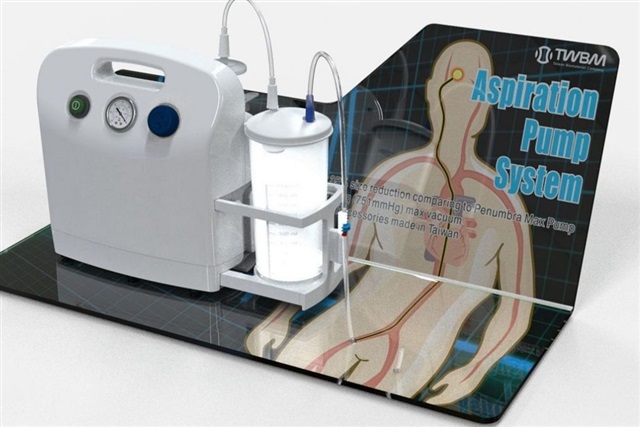
A rare stroke-treatment device is heading to market. Taiwan Biomaterial Company (TWBM), backed by investor Netronix, says its stroke negative pressure pump system catheter guidewire—one of only two such products available worldwide—is slated to begin shipping in the first quarter of 2026.
This specialized catheter guidewire is designed for neurovascular procedures where speed and precision matter. In stroke care, guidewires help specialists navigate fragile brain vessels to reach a blockage. Pairing that access with a negative pressure pump system can support clot-aspiration workflows and streamline mechanical intervention, aiming to reduce procedure time and improve consistency during critical, time-sensitive care.
The announcement underscores how competitive and capacity-constrained this segment is. With so few comparable systems on the global market, a 2026 rollout could help ease supply bottlenecks for hospitals and neurointerventional centers seeking advanced tools for ischemic stroke treatment. It also signals growing momentum from Taiwan’s medtech ecosystem, where companies are investing in minimally invasive devices and next-generation neurovascular technologies.
Key points:
– TWBM plans to start shipments in Q1 2026.
– The stroke negative pressure pump system catheter guidewire is among only two of its kind globally.
– The device targets neurovascular procedures that rely on rapid access and aspiration techniques.
– Netronix is an investor in TWBM, highlighting confidence in the company’s commercialization path.
As the shipment date approaches, expect updates on manufacturing readiness, clinical adoption plans, and training support for interventional teams. For providers and purchasers building stroke-care capabilities, this timeline offers a clear marker for evaluating equipment planning and procurement.